Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (7): 1042-1046.doi: 10.3969/j.issn.2095-4344.2015.07.011
Previous Articles Next Articles
Effects of connective tissue growth factor on wound healing and scarring
He Ze-liang1, Tang Yong2, Yao Zong-jiang1, Zhang Xuan-fen1
- 1Department of Plastic Surgery, 2Department of Critical Diseases, Lanzhou University Second Hospital, Lanzhou 730030, Gansu Province, China
-
Online:2015-02-12Published:2015-02-12 -
Contact:Zhang Xuan-fen, Professor, Department of Plastic Surgery, Lanzhou University Second Hospital, Lanzhou 730030, Gansu Province, China -
About author:He Ze-liang, Studying for master’s degree, Department of Plastic Surgery, Lanzhou University Second Hospital, Lanzhou 730030, Gansu Province, China -
Supported by:the Natural Science Foundation of Gansu Province, No. 1107RJZA207; the Science Research Plan in Health Care Industry of Gansu Province, No. GSWT09~7
CLC Number:
Cite this article
He Ze-liang, Tang Yong, Yao Zong-jiang, Zhang Xuan-fen. Effects of connective tissue growth factor on wound healing and scarring[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(7): 1042-1046.
share this article
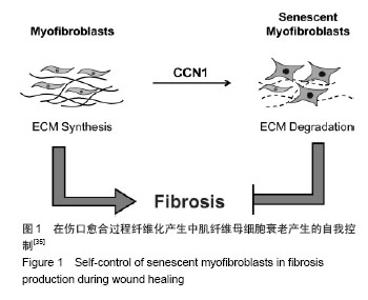
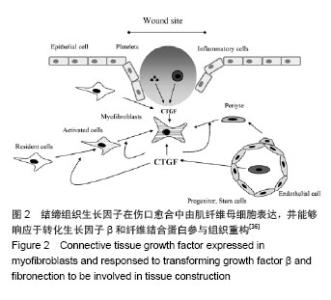
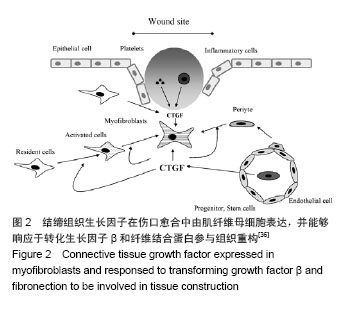
2.1 结缔组织生长因子的定位及来源 CNN2是Bradham等[3]于1991年研究人脐静脉内皮细胞CDNA文库时首次发现的,属于即刻早期基因家族CCN六个成员之一。人结缔组织生长因子基因定位于染色体6q 23.1区,其蛋白含349个氨基酸,相对分子质量为38 000[4]。 结缔组织生长因子主要由皮肤成纤维细胞、肾小球系膜细胞、血管平滑肌细胞、软骨细胞、肝星状细胞等细胞合成分泌[5]。Kapoor等[6]发现皮肤组织修复过程中结缔组织生长因子大部分是由成纤维细胞的特殊类型肌成纤维细胞分泌的,部分周细胞也可以分泌结缔组织生长因子。也有文献显示损伤初始阶段血小板的α颗粒释放可以释放结缔组织生长因子[7]。 结缔组织生长因子在真皮层的含量明显高于表皮 层[8-10]。进一步研究发现正常皮肤表皮层结缔组织生长因子主要定位于黑素细胞[9-10];在真皮乳头层,结缔组织生长因子蛋白与网状纤维结合,分布于真皮层胶原结节处或环绕血管分布,或位于成纤维细胞样细胞的细胞质中,而在更深的真皮层中,结缔组织生长因子蛋白位于真皮成纤维细胞质。 2.2 结缔组织生长因子的生物学作用 结缔组织生长因子可与不同的靶细胞结合,进而介导不同的生物学作用。结缔组织生长因子主要生物学作用有促胚胎发育和有丝分裂,刺激成纤维细胞增殖[5,11];诱导细胞糖蛋白、蛋白聚糖如硫酸乙酰肝素、纤维状胶原如Ⅰ型和Ⅲ型胶原蛋白等细胞外基质产生和沉积[12];刺激内皮细胞基底膜生成和新生血管生成[13-14];介导角质形成细胞和血管内皮细胞的黏附和迁移[14-15];诱导肌成纤维细胞的分化和募集[16-17];加速肉芽组织形成[13];促进并持续纤维化等作用[18-19]。 2.3 结缔组织生长因子在皮肤创面愈合过程中的表达变化 结缔组织生长因子在创面愈合过程中发生动态变化,但其表达规律却没有一致结论。在人体皮肤烧伤创面伤后三四天结缔组织生长因子含量极少,五六天结缔组织生长因子表达略增加,尤其是伤口边缘的表皮层,同时真皮层也发现了少量分布;表达高峰发生于第七八天,毛囊表皮角质形成细胞中表达显著,并在烧伤愈合区域大量存在;伤后11-20 d 结缔组织生长因子的表达再次减少[13]。Rittie等[9]在人体皮肤外伤创面也发现类似结缔组织生长因子的表达趋势,进一步研究还发现伤后24 h 结缔组织生长因子曾发生过显著降低(约55%),在伤后第3天恢复正常,此后逐渐上升至第7天,在基质重塑阶段并没有显著改变。但也有报道在兔耳皮肤外伤创面伤后的前40 d结缔组织生长因子的表达呈持续升高[17]。各观察结果发现结缔组织生长因子的表达时间上具有差异性,这可能与不同动物种类及创面类型有关,但尚未见对照研究种属及创面类型差异时结缔组织生长因子表达区别的文献报道。 此外,有研究显示结缔组织生长因子的表达在急性和慢性创面中也具有差异性[8]:在急性创伤,真皮层的染色相对较弱,而在慢性伤口结缔组织生长因子明显减弱甚至消失,其含量仅约为急性创面的60%。因此,结缔组织生长因子表达减少可能是导致慢性创面发生的原因之一。此外,缺氧、高糖环境及机械应力等也可影响结缔组织生长因子表达变化,三者均可导致结缔组织生长因子的上调[20-21]。 2.4 结缔组织生长因子对创面愈合和瘢痕演变的影响 结缔组织生长因子可能在皮肤创面愈合过程中发挥了重要作用。研究发现恒河猴烧伤创面使用重组人结缔组织生长因子后,创面显著减小,纤维化也更加有序[22]。这主要是由于结缔组织生长因子可加速成纤维细胞增殖和促进包括糖蛋白如纤连蛋白、蛋白聚糖如硫酸乙酰肝素、纤维状胶原如Ⅰ型和Ⅲ型胶原蛋白等细胞外基质合成进而促进肉芽组织形成有关[12,23];还可能与结缔组织生长因子可通过整合素αvβ3介导血管内皮细胞的黏附和迁移并诱导内皮细胞基底膜的产生促进新生血管生成有关[12]。还有研究认为结缔组织生长因子可通过整合素α5β1介导角质形成细胞迁移到创面周围,产生丰富的纤维黏连蛋白促进创面愈合[24]。此外,在大鼠烫伤创面局部外用重组人结缔组织生长因子可缩短愈合时间,并随着结缔组织生长因子浓度的增加其作用呈现递增趋势[25]。这提示结缔组织生长因子可促进创面愈合,且具有浓度依赖性。 结缔组织生长因子作为转化生长因子β的下游产物,除本身的生物学作用外,其介导转化生长因子β的促成纤维细胞增殖、促纤维化、诱导黏附及迁移等作用也是其影响创面愈合的作用机制之一[1,26]。成人皮肤组织中转化生长因子β有转化生长因子β1、β2、β3三个亚型。Colwell等[27]研究证实转化生长因子β1、β2、β3亚型均可刺激结缔组织生长因子的表达。有趣的是,转化生长因子β1可促进创面早期愈合且愈合后持续表达可促进瘢痕生成,而转化生长因子β3却可抑制瘢痕增生[28-30]。因此,结缔组织生长因子介导何种转化生长因子β亚型的作用对于创面愈合的结局具有重要意义。但结缔组织生长因子对于转化生长因子β各亚型的介导作用还需要进一步的研究证实。 另外,小鼠创面应用结缔组织生长因子抗体后创面早期肉芽组织和肉芽组织中血管数量发生减少,晚期肉芽组织含量却没有明显差异[13],这可能与创面中结缔组织生长因子含量降低或作用减弱引起血管形成被抑制以及成纤维细胞增殖和胶原产生减少有关,也提示结缔组织生长因子在皮肤创面愈合中的作用可能仅限于早期阶段,因结缔组织生长因子主要作用于成纤维细胞,而成纤维细胞在创面愈合的早期增殖旺盛,晚期增殖减缓。然而,结缔组织生长因子基因敲除小鼠伤后第7天创面愈合情况、基质沉积及成纤维细胞增殖与对照组没有明显差异[16],且外用结缔组织生长因子反义寡核苷酸并没有显著改变肉芽组织含量及伤口愈合[17],这提示结缔组织生长因子在创面愈合过程中可能没有发挥关键作用,抑或与结缔组织生长因子对创面愈合影响研究中使用的动物模型、结缔组织生长因子及其抑制剂的量不能完全发挥作用有关。 结缔组织生长因子在瘢痕疙瘩和增生性瘢痕中含量较正常皮肤和瘢痕组织明显高表达,尤其是在瘢痕疙瘩临近组织中也发现了结缔组织生长因子的高表达,提示可能与瘢痕疙瘩的浸润性生长也有关系[10,31]。借鉴辛伐他汀可降低结缔组织生长因子的表达抑制心脏纤维化的现象,有研究将其应用于皮肤瘢痕组织中发现可抑制瘢痕增生[32]。尽管外用结缔组织生长因子反义寡核苷酸肉芽组织含量及伤后愈合并没有显著改变,但是伤口后期处理较早期给药可更好地减少瘢痕增生,且伴随着Ⅰ型及Ⅲ型胶原蛋白、基质金属蛋白酶的组织抑制剂、α-平滑肌肌动蛋白表达阳性的肌成纤维细胞显著减少[17]。结缔组织生长因子可能参与增生性瘢痕中肌成纤维细胞的分化或者招募,增加α-平滑肌肌动蛋白的表达,而α-平滑肌肌动蛋白表达阳性肌成纤维细胞的持续存在是导致细胞外基质沉积、伤口收缩和瘢痕挛缩形成的因素之一[33-34](见图1)。当细胞外基质过量分泌时,细胞外基质成分和细胞间不能形成有机排列的正常结构,最终纤维化、瘢痕形成[35-37](见图2)。这表明结缔组织生长因子可通过促进肌成纤维细胞的分化或招募参与病理性瘢痕的发生。此外,结缔组织生长因子还可通过整合素α5β1介导角质形成细胞的黏附加速上皮愈合,而上皮愈合延迟也是导致创面慢性化甚至病理性瘢痕发生的原因[14]。 "

| [1]Penn JW, Grobbelaar AO, Rolfe KJ. The role of the TGF-beta family in wound healing, burns and scarring: a review. Int J Burns Trauma.2012;2(1):18-28.
[2]Shi-wen X, Pennington D, Holmes A, et al. Autocrine overexpression of CTGF maintains fibrosis: RDA analysis of fibrosis genes in systemic sclerosis. Exp Cell Res.2000; 259(1):213-224.
[3]Bradham DM, Igarashi A, Potter RL, et al. Connective tissue growth factor: a cysteine-rich mitogen secreted by human vascular endothelial cells is related to the SRC-induced immediate early gene product CEF-10. J Cell Biol. 1991; 114(6):1285-1294.
[4]胡振富,高建华,李薇,等.瘢痕疙瘩的差异表达基因谱研究[J].南方医科大学学报,2006,26(3):308-312.
[5]向小燕,岑瑛.结缔组织生长因子与病理性瘢痕[J]. 西部医学, 2005,17(2):178-180.
[6]Kapoor M, Liu S, Huh K, et al. Connective tissue growth factor promoter activity in normal and wounded skin. Fibrogenesis Tissue Repair.2008;1(1):3.
[7]Chen CC, Lau LF. Functions and mechanisms of action of CCN matricellular proteins. Int J Biochem Cell Biol.2009; 41(4):771-783.
[8]Minhas U, Martin TA, Ruge F, et al. Pattern of expression of CCN family members Cyr61, CTGF and NOV in human acute and chronic wounds.Exp Ther Med.2011;2(4):641-645.
[9]Rittie L, Perbal B, Castellot JJ, et al. Spatial-temporal modulation of CCN proteins during wound healing in human skin in vivo. J Cell Commun Signal. 2011;5(1):69-80.
[10]王勇,王少华,唐胜建,等.病理性瘢痕中结缔组织生长因子的表达及其意义[J].中国美容医学, 2008,17(11):1628-1630.
[11]Ivkovic S, Yoon BS, Popoff SN, et al. Connective tissue growth factor coordinates chondrogenesis and angiogenesis during skeletal development. Development. 2003;130(12): 2779-2791.
[12]Chaqour B. Molecular control of vascular development by the matricellular proteins CCN1 (Cyr61) and CCN2 (CTGF). Trends Dev Biol.2013;7:59-72.
[13]Alfaro MP, Deskins DL, Wallus M, et al. A physiological role for connective tissue growth factor in early wound healing. Lab Invest.2013;93(1):81-95.
[14]Mustoe TA. Scars and Keloids:Several treatments are used,but the evidence base is lacking. BMJ.2004;328(7452): 1329.
[15]Kiwanuka E, Andersson L, Caterson EJ, et al. CCN2 promotes keratinocyte adhesion and migration via integrin alpha5beta1. Exp Cell Res. 2013;319(19):2938-2946.
[16]Liu S, Thompson K, Leask A. CCN2 expression by fibroblasts is not required for cutaneous tissue repair. Wound Repair Regen.2014;22(1):119-124.
[17]Sisco M, Kryger ZB, O'Shaughnessy KD, et al. Antisense inhibition of connective tissue growth factor (CTGF/CCN2) mRNA limits hypertrophic scarring without affecting wound healing in vivo. Wound Repair Regen.2008;16(5):661-673.
[18]Bonniaud P, Martin G, Margetts PJ, et al. Connective tissue growth factor is crucial to inducing a profibrotic environment in "fibrosis-resistant" BALB/c mouse lungs. Am J Respir Cell Mol Biol.2004;31(5):510-516.
[19]Leask A. Transcriptional profiling of the scleroderma fibroblast reveals a potential role for connective tissue growth factor (CTGF) in pathological fibrosis. Keio J Med.2004;53(2):74-77.
[20]Geisinger MT, Astaiza R, Butler T, et al. Ets-1 is essential for connective tissue growth factor (CTGF/CCN2) induction by TGF-beta1 in osteoblasts. PLoS One. 2012;7(4):e35258.
[21]Daniels A, Van Bilsen M, Goldschmeding R, et al. Connective tissue growth factor and cardiac fibrosis. Acta physiologica. 2009;195(3):321-338.
[22]Liu LD, Shi HJ, Jiang L, et al. The repairing effect of a recombinant human connective-tissue growth factor in a burn-wounded rhesus-monkey (Macaca mulatta) model. Biotechnol Appl Biochem.2007;47(Pt 2):105-112.
[23]Czubryt MP. Common threads in cardiac fibrosis, infarct scar formation, and wound healing. Fibrogenesis Tissue Repair. 2012;5(1):19.
[24]Kiwanuka E, Andersson L, Caterson EJ, et al. CCN2 promotes keratinocyte adhesion and migration via integrin alpha5beta1.Exp Cell Res. 2013;319(19):2938-2946.
[25]谢正福,余书勤.重组人结缔组织生长因子(rhCTGF)促进大鼠烫伤创面愈合的实验研究[J]. 广东药学院学报, 2005,21(2):178-182.
[26]Ponticos M. Connective tissue growth factor (CCN2) in blood vessels. Vascul Pharmacol.2013;58(3):189-193.
[27]Colwell AS, Phan TT, Kong W, et al. Hypertrophic scar fibroblasts have increased connective tissue growth factor expression after transforming growth factor-beta stimulation. Plast Reconstr Surg.2005;116(5):1387-1390, 1391-1392.
[28]Wehrhan F, Rodel F, Grabenbauer GG, et al. Transforming growth factor beta 1 dependent regulation of Tenascin-C in radiation impaired wound healing. Radiother Oncol.2004; 72(3):297-303.
[29]Fan MS, Jiang ZY, Zou YF, et al. [Effect of transforming growth factor beta1 on the expression of matrix metalloproteinase 9, tissue inhibitor of metalloproteinase 1 and nuclear factor kappa B signalling pathway in the human amniotic cells WISH]. Zhonghua Fu Chan Ke Za Zhi. 2013; 48(1):29-33.
[30]Ohno S, Hirano S, Kanemaru S, et al. Transforming growth factor beta3 for the prevention of vocal fold scarring. Laryngoscope.2012;122(3):583-589.
[31]Igarashi A, Nashiro K, Kikuchi K, et al. Connective tissue growth factor gene expression in tissue sections from localized scleroderma, keloid, and other fibrotic skin disorders. J Invest Dermatol.1996;106(4):729-733.
[32]Mun JH,Kim YM,Kim BS,et al.Simvastatin inhibits transforming growth factor-beta1-induced expression of type I collagen, CTGF, and alpha-SMA in keloid fibroblasts. Wound Repair Regen.2014;22(1):125-133.
[33]Lee CH, Shah B, Moioli EK, et al. CTGF directs fibroblast differentiation from human mesenchymal stem/stromal cells and defines connective tissue healing in a rodent injury model. J Clin Invest.2010;120(9):3340-3349.
[34]Wynn TA.Cellular and molecular mechanisms of fibrosis.J pathol. 2008;214(2):199-210.
[35]Jun JI, Lau LF. Cellular senescence controls fibrosis in wound healing. Aging (Albany NY).2010;2(9):627-631.
[36]Shi-Wen X, Leask A, Abraham D. Regulation and function of connective tissue growth factor/CCN2 in tissue repair, scarring and fibrosis. Cytokine & growth factor reviews. 2008; 19(2): 133-144.
[37]Hall-Glenn F, Lyons K M. Roles for CCN2 in normal physiological processes. Cellular and Molecular Life Sciences. 2011;68(19): 3209-3217. |
| [1] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [2] | Liu Fang, Shan Zhengming, Tang Yulei, Wu Xiaomin, Tian Weiqun. Effects of hemostasis and promoting wound healing of ozone sustained-release hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3445-3449. |
| [3] | Chen Zhenyu, Zhang Xiaoning, Luo Yuxin, Liang Jianwei, Yan Chi. Evaluation of silk fibroin/curcumin composite film for promoting wound healing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2554-2561. |
| [4] | Zhao Xin, Shi Xin, Chen Bei, Cao Yanpeng, Chen Yaowu, Liu Xiaoren, He Yusheng, He Liyun, Li Xiying, Liu Jun. Vacuum sealing drainage enhances wound healing by up-regulating collagen type I/III ratio in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(32): 5122-5127. |
| [5] | Li Qinwen, Liang Jie, Wang Dongmei, Shang Zhenghui. Fibrotic changes in rat dorsal root ganglion following chronic sciatic nerve compression [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4686-4691. |
| [6] | Liu Yanhua, Zhu Zhou, Wan Qianbing. A drug-loading system for electrospinning wound repair: component selection and construction strategy [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4465-4473. |
| [7] |
Geng Kang, Ding Xiaobin, Tian Xinli, Wang Xue, Yang Yuting, Yan Hong.
Electrical stimulation promotes wound healing and angiogenesis in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4152-4156. |
| [8] |
Gu Yingxuan, Huang Linfeng, Hu Xiaohui, Quan Xiaoming, Kang Liangqi, Zhou Linghan, Wang Xiaojun.
Platelet-rich plasma combined with negative pressure for chronic refractory wounds: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4257-4264. |
| [9] | Liang Zhou, Wen Lichun, He Zhong, Huang Zheng, Li Kaijing, Yang Xiaoping, Liang Shihe, Pang Gexiong. Three-dimensional printing combined with virtual surgical design in the treatment of complex Pilon fractures [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(24): 3786-3791. |
| [10] | Zhong Shuxian, Shi Yuqing, Yang Yalan, Li Chun. Effect of soft silicone silver ion dressing applied in burn wounds [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3602-3608. |
| [11] | Zhong Shuxian, Yang Yalan, Shi Yuqing, Li Chun . Meta-analysis of platelet-rich plasma in repairing burn wounds [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(14): 2291-2296. |
| [12] | Zhou Yanxing1, Peng Xinsheng2, Hou Gan1, Li Jiangbin1, Zhang Hua1, Zhou Zhikun2, Zhou Yanfang3 . Inhibitory effect of capsaicin on fibroblast proliferation and its molecular mechanism [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(7): 1018-1022. |
| [13] | Yuan Yacui1, Liu Linna2, Liu Xiwen3, Li Yuejun1, Zhao Congying1, Han Lin1. Evaluation of the effectiveness and safety of closed negative pressure drainage nursing system (PU suction cup type): a perspective, randomized, open, parallel controlled and non-inferiority trial [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5676-5681. |
| [14] | Ma Hongwei, Liu Panyun, Zhang Yaqiong. Ginsenoside-induced bone marrow mesenchymal stem cell intervention can affect healing and Wnt/beta-catenin signaling in rats with diabetic skin ulcer [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(33): 5300-5306. |
| [15] | Li Weiming, Sun Dali, Li Yijun, Li Shumin, Sun Yanbo, Cui Jin, Xu Pengyuan, Xu Qingwen. Deep seawater promotes cutaneous wound healing in diabetic mice via activating Wnt/β-catenin pathway [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(31): 5017-5022. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||